Electrochemistry Class 12 Notes + PDF Download + NCERT Solutions
Classroom Notes — Hindi + English (द्विभाषी)
Class 12 Chemistry | NCERT Unit 2 | NEET · JEE · CBSE Board
इस अध्याय में हम Electrochemistry Class 12 Notes को बहुत ही सरल और आसान भाषा में समझेंगे। यहाँ पर हम आपको PDF notes for quick revision, audio notes जिससे आप सुनकर भी पढ़ सकते हैं, practice के लिए MCQ questions, और fill in the blanks उपलब्ध करवा रहे हैं। इसके साथ ही आपकी exam preparation को बेहतर बनाने के लिए important FAQ questions भी शामिल किए गए हैं। ये सभी सामग्री आपको chapter को अच्छे से समझने और Class 12 board exam, CBSE board, अन्य state boards के साथ-साथ NEET, IIT-JEE जैसे competitive exams की तैयारी को मजबूत बनाने में मदद करेंगी।
-
Electrochemistry Class 12 Notes + PDF Download + NCERT Solutions– Introduction
Electrochemistry रसायन विज्ञान की वह शाखा है जो chemical reactions और electrical energy के बीच संबंध का अध्ययन करती है।
Electrochemistry वह topic है जिसमें हम समझते हैं कि chemical reaction से electricity कैसे बनती है और electricity की मदद से नए chemical reactions कैसे कराए जाते हैं।
दो प्रकार के Cells (Two Types of Cells)
|
Feature |
Galvanic / Voltaic Cell |
Electrolytic Cell |
|
Energy conversion |
Chemical → Electrical |
Electrical → Chemical |
|
Reaction type |
Spontaneous (ΔG < 0) |
Non-spontaneous (ΔG > 0) |
Teacher–Student Dialogue
|
Sir (Teacher) बच्चों, एक simple सवाल — आपके घर में जो torch या TV remote में battery लगाते हो — वो काम कैसे करती है? |
|
Student – Priya Sir, battery में chemical reaction होती है जिससे current निकलती है? |
|
Sir (Teacher) बिल्कुल सही! यही Electrochemistry है। |
Types of Electrochemical Cells
Local Example: Gaon ki dukaan mein jo ‘Eveready’ cell milti hai — usme bhi yahi chemistry hai! | सेब काटने के बाद brown होना = spontaneous! | Iron में rust = spontaneous!
Key Points — याद रखो!
|
1 |
Galvanic/Voltaic Cell — Chemical energy → Electrical energy (spontaneous redox reaction) |
|
2 |
Electrolytic Cell — Electrical energy → Chemical reaction (non-spontaneous) |
|
3 |
Electrochemistry is used industrially to make Cl₂, NaOH, metals like Na, Mg, Al |
|
4 |
Batteries और Fuel Cells — galvanic cells हैं |
-
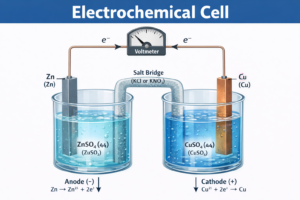
Galvanic Cell – Daniell Cell
A galvanic cell is an electrochemical cell that converts chemical energy of a spontaneous redox reaction into electrical energy.
Daniell Cell — संरचना (Structure)
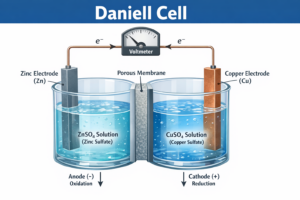
Cell Notation: Zn(s) | Zn²⁺(aq) || Cu²⁺(aq) | Cu(s)
Overall Reaction: Zn(s) + Cu²⁺(aq) → Zn²⁺(aq) + Cu(s) E°cell = 1.10 V
Half Reactions — अर्ध अभिक्रियाएँ
|
Electrode |
Half Reaction |
Type |
|
Anode (Zinc −) |
Zn(s) → Zn²⁺(aq) + 2e⁻ |
Oxidation |
|
Cathode (Copper +) |
Cu²⁺(aq) + 2e⁻ → Cu(s) |
Reduction |
Classroom Dialogue
|
Sir (Teacher) Daniell Cell में दो beakers हैं। Left में Zinc electrode — ZnSO₄ solution में। Right में Copper electrode — CuSO₄ solution में। |
|
Student – Arjun Sir, Salt Bridge क्यों लगाते हैं? |
|
Sir (Teacher) Salt Bridge दो काम करता है: |
Salt bridge mein KCl ya KNO₃ hota hai — same namak jo aapke kitchen mein hai! | 🔋 Memory Trick: ‘Red Cat, An Ox’ — Reduction at Cathode, Oxidation at Anode!
What is a salt bridge?
Student: asked Priya
Teacher: Salt Bridge (सॉल्ट ब्रिज) एक ऐसी नली (tube) होती है जो दो अलग-अलग घोलों (solutions) को जोड़ती है और इलेक्ट्रॉनों के प्रवाह को संतुलित रखने में मदद करती है। इसे आमतौर पर electrochemical cell/galvanic cell में उपयोग किया जाता है।
|
Student – Priya Sir, Anode और Cathode में difference क्या है? |
|
👨🏫 Sir (Teacher) Memory Trick: |
Key Points
|
1 |
Anode: Oxidation (negative in galvanic cell) | Zn → Zn²⁺ + 2e⁻ |
|
2 |
Cathode: Reduction (positive in galvanic cell) | Cu²⁺ + 2e⁻ → Cu |
|
3 |
Electrons flow: Anode → external wire → Cathode |
|
4 |
Current direction: Cathode → Anode (opposite to electrons) |
|
5 |
Cell notation: Zn(s) | Zn²⁺(aq) || Cu²⁺(aq) | Cu(s) [ | = phase boundary, || = salt bridge ] |
|
🩺 NEET Tip Q: In a Daniell cell, which electrode acts as anode? |
-
Standard Electrode Potential & EMF
Standard Electrode Potential (E°): When concentrations of all species in a half-cell are unity, the electrode potential is called standard electrode potential. Standard Hydrogen Electrode (SHE) is assigned E° = 0.00 V as reference.
E°cell = E°cathode − E°anode = E°right − E°left
ΔG° = −nFE°cell | ΔG° = −RT ln Kc
Table 2.1 — Standard Electrode Potentials at 298 K (Selected)
|
Half-Reaction (Reduction) |
E°/V |
Notes |
|
F₂(g) + 2e⁻ → 2F⁻ |
+2.87 |
Strongest Oxidising Agent |
|
Au³⁺ + 3e⁻ → Au |
+1.40 |
Gold — no rust! |
|
Cl₂ + 2e⁻ → 2Cl⁻ |
+1.36 |
|
|
O₂ + 4H⁺ + 4e⁻ → 2H₂O |
+1.23 |
|
|
Ag⁺ + e⁻ → Ag |
+0.80 |
|
|
Cu²⁺ + 2e⁻ → Cu |
+0.34 |
Cathode in Daniell cell |
|
2H⁺ + 2e⁻ → H₂ |
0.00 |
SHE — Reference |
|
Zn²⁺ + 2e⁻ → Zn |
−0.76 |
Anode in Daniell cell |
|
Mg²⁺ + 2e⁻ → Mg |
−2.36 |
|
|
Li⁺ + e⁻ → Li |
−3.05 |
Strongest Reducing Agent |
Daniell Cell — E°cell Calculation
|
👨🏫 Sir (Teacher) Formula याद है? |
💡 Gold (सोना) E° = +1.40 V — इसीलिए gold rust नहीं करता! | Zinc E° = −0.76 V — इसीलिए iron को galvanize करने में use होता है (Galvanized iron = zinc coating)!
|
⚙️ JEE Important — ΔG° Calculation ΔG° = −nFE°cell |
-
Nernst Equation — नर्न्स्ट समीकरण
Nernst equation gives the electrode potential at any concentration with respect to the standard hydrogen electrode:
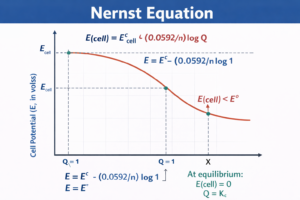
E(cell) = E°(cell) − (RT/nF) × ln Q
At 298 K: E(cell) = E°(cell) − (0.0592/n) × log Q
For Daniell Cell: E(cell) = E°(cell) − (0.0296) × log [Zn²⁺]/[Cu²⁺]
Triangle of Relations — EMF, ΔG°, Kc
E°cell = (0.059/n) log Kc | ΔG° = −nFE°cell = −RT ln Kc
Classroom Dialogue
|
👨🏫 Sir (Teacher) अब तक हमने assume किया था कि concentration = 1 M है। Real life में ऐसा नहीं होता। |
🔋 Battery discharge होते समय: Zn²⁺ बढ़ता है, Cu²⁺ घटता है → Q बढ़ता है → Voltage कम होती जाती है। यही कारण है कि पुरानी battery का voltage कम हो जाता है!
Example 2.1 — Solved Step-by-Step
|
📝 Example 2.1 Cell: Mg | Mg²⁺(0.130 M) || Ag⁺(0.0001 M) | Ag |
Equilibrium Constant from Nernst Equation
At equilibrium: E(cell) = 0
0 = E°(cell) − (0.059/n) × log Kc
E°(cell) = (0.059/n) × log Kc
For Daniell cell (E° = 1.10 V, n = 2):
log Kc = (1.10 × 2) / 0.059 = 37.288
Kc = 2 × 10³⁷ at 298 K
-
Conductance of Electrolytic Solutions— चालकता
Important Formulas
|
Quantity |
Formula / Expression |
|
Resistance |
R = ρ × (l/A) | Unit: Ohm (Ω) |
|
Conductanceκ |
G = 1/R = κ × (A/l) | Unit: Siemens (S) |
|
Conductivity |
K= G × (l/A) where
|
|
Molar Conductivity |
Λm = κ/c | Unit: S m² mol⁻¹ or S cm² mol⁻¹ |
|
Cell Constant |
G* = l/A | Dimension: length⁻¹ |
|
Kohlrausch Law |
Λ°m = ν₊ λ°₊ + ν₋ λ°₋ |
|
Strong Electrolytes |
Λm = Λ°m − A√c (Debye-Hückel-Onsager) |
|
Degree of Dissociation |
α = Λm / Λ°m |
|
Ka for weak acid |
Ka = cα² / (1−α) |
👨🏫 Classroom Dialogue — Conductance
|
👨🏫 Sir (Teacher) पहले समझो: electricity कैसे flow करती है? |
💡 नमक के पानी में (salt water) current चलती है — यही ionic conductance है! | 🍬 Cheeni (sugar) dissolve → no ions → no current! Namak (NaCl) dissolve → ions → current chalti hai!
Kohlrausch Law — Application for Weak Electrolytes
|
📝 Example 2.8 — Acetic Acid Λ°m(CH₃COOH) = Λ°m(HCl) + Λ°m(CH₃COONa) − Λ°m(NaCl) |
|
🩺 NEET/JEE Tip Most asked: Degree of dissociation (α) and Ka from conductance data. |
-
Electrolysis and Faraday’s Laws — विद्युत अपघटन
In an electrolytic cell, external source of voltage is used to bring about a chemical reaction. Electrolysis = using external current to drive non-spontaneous reactions.
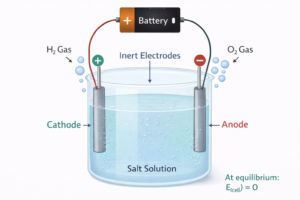
Faraday’s Laws of Electrolysis
|
⚡ Faraday’s First Law The amount of chemical reaction which occurs at any electrode during electrolysis is proportional to the quantity of electricity passed through the electrolyte. |
|
Faraday’s Second Law Amounts of different substances liberated by the same quantity of electricity are proportional to their chemical equivalent weights. |
Example 2.10 — Solved
|
CuSO₄ Electrolysis Given: t = 10 min = 600 s, I = 1.5 A |
Key Points
|
1 |
Cathode = reduction | Anode = oxidation (same as galvanic cell) |
|
2 |
1 Faraday = 96487 C mol⁻¹ = charge of 1 mol electrons |
|
3 |
w = (M/nF) × I × t — Faraday’s First Law |
|
4 |
Inert electrodes (Pt, Au) — don’t participate; Reactive electrodes — do participate |
|
5 |
Aqueous NaCl electrolysis → H₂ at cathode, Cl₂ at anode, NaOH in solution |
Gold/Silver jewellery की coating — यही electroplating है! | 🔩 Copper refining — impure Cu anode, pure Cu cathode, CuSO₄ electrolyte!
-
Batteries, Fuel Cells and Corrosion
Primary Batteries (Non-rechargeable)
|
Feature |
Dry Cell (Leclanche) |
Mercury Cell |
|
Anode |
Zn (container) |
Zn-Hg amalgam |
|
Cathode |
Graphite rod in MnO₂ |
HgO + Carbon paste |
|
Electrolyte |
NH₄Cl + ZnCl₂ paste |
KOH + ZnO paste |
|
Voltage |
~1.5 V |
1.35 V (constant) |
|
Use |
Torch, remote, clocks |
Hearing aids, watches |
Lead Storage Battery (Secondary)
Anode: Pb(s) + SO₄²⁻(aq) → PbSO₄(s) + 2e⁻
Cathode: PbO₂(s) + SO₄²⁻(aq) + 4H⁺(aq) + 2e⁻ → PbSO₄(s) + 2H₂O(l)
Overall: Pb(s) + PbO₂(s) + 2H₂SO₄(aq) → 2PbSO₄(s) + 2H₂O(l)
Electrolyte: 38% H₂SO₄ solution | Voltage: ~2 V per cell (car battery = 6 cells = 12 V)
जब गाड़ी चलती है — alternator से current आती है और battery recharge होती है। Recharging reverses the reaction: PbSO₄ → Pb and PbO₂.
Fuel Cell — H₂–O₂
Cathode: O₂(g) + 2H₂O(l) + 4e⁻ → 4OH⁻(aq)
Anode: 2H₂(g) + 4OH⁻(aq) → 4H₂O(l) + 4e⁻
Overall: 2H₂(g) + O₂(g) → 2H₂O(l)
Efficiency: ~70% (thermal power plants = 40%) | Used in Apollo space programme!
Corrosion — Electrochemical Process
Anode: 2Fe(s) → 2Fe²⁺(aq) + 4e⁻ E° = −0.44 V
Cathode: O₂(g) + 4H⁺(aq) + 4e⁻ → 2H₂O(l) E° = +1.23 V
Overall: 2Fe(s) + O₂(g) + 4H⁺(aq) → 2Fe²⁺(aq) + 2H₂O(l) E°cell = 1.67 V
Fe²⁺ is further oxidised by O₂ to form hydrated ferric oxide (Fe₂O₃·xH₂O) = Rust
|
Prevention of Corrosion ( Corrosion को रोकने के कुछ तरीके हैं:) 1. Metal पर paint या coating करना (e.g., bisphenol) |
Bridge, ship, railway tracks — iron rust रोकने के लिए zinc/magnesium sacrificial anodes लगाते हैं। India में crores ka nuksaan hota hai rust se har saal!
-
Exam Summary and Important Formulas — परीक्षा के लिए Summary
All Important Formulas at a Glance
|
Quantity |
Formula |
Meaning |
|
E°cell |
E°cathode − E°anode |
Standard cell potential |
|
ΔG° |
−nFE°cell |
Standard Gibbs energy |
|
ΔG° |
−RT ln Kc |
Gibbs from equilibrium constant |
|
E°cell |
(0.059/n) × log Kc |
E° from Kc at 298 K |
|
Nernst |
E = E° − (0.059/n) × log Q |
Cell potential at any conc. |
|
Conductance |
G = 1/R = κA/l |
From resistance |
|
Molar Cond. |
Λm = κ/c |
From conductivity |
|
Kohlrausch |
Λ°m = ν₊λ°₊ + ν₋λ°₋ |
Limiting molar conductivity |
|
Debye law |
Λm = Λ°m − A√c |
Strong electrolytes |
|
Dissociation |
α = Λm/Λ°m |
Degree of dissociation |
|
Ka |
cα²/(1−α) |
Dissociation constant |
|
Faraday 1st |
w = MIt/nF |
Mass deposited in electrolysis |
|
Charge |
Q = It |
Coulombs = Ampere × seconds |
Exam Strategy — परीक्षा की तैयारी
|
CBSE Board Focus areas: |
|
NEET Focus areas: |
|
JEE Focus areas: |
Electrochemistry Class 12 MCQ Questions
SECTION 1 — Multiple Choice Questions (MCQ)
30 Questions — NEET | IIT JEE | CBSE | State Boards
NEET Q1. In a Daniell cell, at the anode the reaction that takes place is:
(A) Cu(s) → Cu²⁺(aq) + 2e⁻
(B) Zn(s) → Zn²⁺(aq) + 2e⁻
(C) Cu²⁺(aq) + 2e⁻ → Cu(s)
(D) Zn²⁺(aq) + 2e⁻ → Zn(s)
✅ Answer: (B) Zn(s) → Zn²⁺(aq) + 2e⁻
Explanation: Zn is the anode. Oxidation always happens at the anode. Zinc loses two electrons and goes into solution as Zn²⁺ ions. याद रखो — AN OX = Anode पर Oxidation।
CBSE Q2. The standard electrode potential (E°) of the Standard Hydrogen Electrode is:
(A) 1.00 V
(B) 0.00 V
(C) −1.00 V
(D) +0.76 V
✅ Answer: (B) 0.00 V
Explanation: By international convention, SHE is assigned exactly 0.00 V. It is the universal reference electrode. अन्तर्राष्ट्रीय convention के अनुसार SHE = 0.00 V।
JEE Q3. Which of the following has the highest standard reduction potential?
(A) Zn²⁺/Zn
(B) Cu²⁺/Cu
(C) F₂/F⁻
(D) Li⁺/Li
✅ Answer: (C) F₂/F⁻
Explanation: Fluorine (F₂) has the highest standard reduction potential of +2.87 V — it is the strongest oxidising agent. फ्लुओरीन सबसे strong oxidising agent है।
NEET Q4. The cell potential E°cell for the Daniell cell (E°Cu = +0.34 V, E°Zn = −0.76 V) is:
(A) +0.42 V
(B) +1.10 V
(C) −1.10 V
(D) −0.42 V
✅ Answer: (B) +1.10 V
Explanation: E°cell = E°cathode − E°anode = 0.34 − (−0.76) = +1.10 V. Positive value = spontaneous reaction. धनात्मक E°cell = अभिक्रिया स्वतः होगी।
CBSE Q5. A salt bridge in a galvanic cell:
(A) Increases the EMF of the cell
(B) Supplies electrons to complete the circuit
(C) Maintains electrical neutrality in both half-cells
(D) Acts as a cathode
✅ Answer: (C) Maintains electrical neutrality in both half-cells
Explanation: A salt bridge (KCl or KNO₃ in agar gel) allows ion flow between half-cells, maintaining electrical neutrality and completing the circuit. Salt bridge बिना cell काम नहीं करेगा।
JEE Q6. According to Nernst equation, the cell potential E at 298 K is given by:
(A) E = E° + (0.059/n) log Q
(B) E = E° − (RT/nF) log Q
(C) E = E° − (0.059/n) log Q
(D) E = E° × (0.059/n) log Q
✅ Answer: (C) E = E° − (0.059/n) log Q
Explanation: At 298 K: E = E° − (0.0592/n) log Q. As Q increases, E decreases. Q बढ़ने पर E घटता है।
NEET Q7. The relation between standard Gibbs energy change and standard cell potential is:
(A) ΔG° = nFE°cell
(B) ΔG° = −nFE°cell
(C) ΔG° = nF/E°cell
(D) ΔG° = −nF/E°cell
✅ Answer: (B) ΔG° = −nFE°cell
Explanation: ΔG° = −nFE°cell. Positive E°cell → Negative ΔG° → Spontaneous. यह तीनों (E°, ΔG°, Kc) आपस में जुड़े हैं।
CBSE Q8. Which of the following is a secondary battery?
(A) Dry cell (Leclanche cell)
(B) Mercury cell
(C) Lead storage battery
(D) Hydrogen-oxygen fuel cell
✅ Answer: (C) Lead storage battery
Explanation: A secondary battery can be recharged. Lead storage battery is the most common example — used in cars. Lead battery को recharge किया जा सकता है।
JEE Q9. The SI unit of molar conductivity (Λm) is:
(A) S cm⁻¹
(B) S m⁻¹
(C) S m² mol⁻¹
(D) S cm² mol⁻¹
✅ Answer: (C) S m² mol⁻¹
Explanation: Λm = κ/c. If κ is in S m⁻¹ and c in mol m⁻³, then Λm = S m² mol⁻¹. SI unit याद रखो — S m² mol⁻¹।
NEET Q10. For a weak electrolyte, as dilution increases, molar conductivity:
(A) Decreases slowly
(B) Remains constant
(C) Increases sharply and approaches limiting value
(D) First increases then decreases
✅ Answer: (C) Increases sharply and approaches limiting value
Explanation: Dilution increases dissociation of weak electrolytes → more ions → sharp increase in Λm. Weak electrolyte dilute होने पर ions बढ़ते हैं।
CBSE Q11. Kohlrausch’s law states that at infinite dilution, molar conductivity of an electrolyte is:
(A) Sum of conductivities of the solvent
(B) Sum of individual ionic conductivities of its ions
(C) Product of ionic conductivities
(D) Equal to its specific conductance
✅ Answer: (B) Sum of individual ionic conductivities of its ions
Explanation: Λ°m = ν⁺λ°⁺ + ν⁻λ°⁻. Each ion migrates independently at infinite dilution. अनंत dilution पर ions स्वतंत्र रूप से migrate करते हैं।
NEET Q12. In electrolysis, the quantity of electricity required to deposit one mole of a monovalent metal is:
(A) 96487/2 C
(B) 96487 C
(C) 2 × 96487 C
(D) 96487 × 3 C
✅ Answer: (B) 96487 C
Explanation: 1 Faraday = 96487 C = charge of 1 mole of electrons. For M⁺ → M, only 1F needed. एकसंयोजी धातु के लिए 1 Faraday चाहिए।
JEE Q13. The process of rusting of iron is:
(A) A physical process
(B) A reversible electrochemical process
(C) An irreversible electrochemical process
(D) A simple chemical process with no electron transfer
✅ Answer: (C) An irreversible electrochemical process
Explanation: Rusting = an electrochemical process. Iron = anode (oxidation), O₂ = cathode (reduction). यह irreversible है। जंग लगना एक electrochemical प्रक्रिया है।
CBSE Q14. The direction of electron flow in a galvanic cell is from:
(A) Cathode to anode through the external circuit
(B) Anode to cathode through the external circuit
(C) Anode to cathode through the salt bridge
(D) Cathode to anode through the solution
✅ Answer: (B) Anode to cathode through the external circuit
Explanation: Electrons are produced at anode (oxidation) and flow through wire to cathode (reduction). Current flows opposite to electrons. Electrons anode से cathode की ओर बाहरी तार से जाते हैं।
NEET Q15. Which metal CANNOT be stored in a zinc container?
(A) ZnSO₄ solution
(B) CuSO₄ solution
(C) MgSO₄ solution
(D) Na₂SO₄ solution
✅ Answer: (B) CuSO₄ solution
Explanation: Zinc (E° = −0.76 V) is more reactive than copper (E° = +0.34 V), so Zn would displace Cu from CuSO₄. Zinc जो ज़्यादा reactive है वो Copper को displace कर देगा।
JEE Q16. At equilibrium in a galvanic cell, the cell potential E is:
(A) Equal to E°cell
(B) Greater than E°cell
(C) Zero
(D) Negative
✅ Answer: (C) Zero
Explanation: At equilibrium, Q = Kc. Nernst equation → E = 0. No net current flows. साम्यावस्था पर E = 0 हो जाता है।
CBSE Q17. Limiting molar conductivity (Λ°m) for weak electrolytes is determined by:
(A) Direct extrapolation of conductivity vs concentration graph
(B) Using Kohlrausch law and Λ°m values of strong electrolytes
(C) Measuring at exactly 1 M concentration
(D) Using the Debye-Hückel equation
✅ Answer: (B) Using Kohlrausch law and Λ°m values of strong electrolytes
Explanation: Weak electrolytes show non-linear graph near zero; Kohlrausch’s law is used instead. Weak electrolytes के लिए direct extrapolation संभव नहीं।
NEET Q18. The standard electrode potential of a cell is related to equilibrium constant Kc by:
(A) E°cell = (nF/RT) log Kc
(B) E°cell = (0.059/n) log Kc at 298 K
(C) E°cell = (RT/nF) log Kc
(D) E°cell = n × 0.059 × log Kc
✅ Answer: (B) E°cell = (0.059/n) log Kc at 298 K
Explanation: At equilibrium, E = 0, Q = Kc. From Nernst: E°cell = (0.059/n) log Kc at 298 K. E° और Kc का relationship।
CBSE Q19. Which of the following is used as a fuel in hydrogen-oxygen fuel cells?
(A) Coal
(B) Petrol
(C) Hydrogen gas
(D) Methane
✅ Answer: (C) Hydrogen gas
Explanation: In H₂-O₂ fuel cell: H₂ at anode, O₂ at cathode → water + electricity (~70% efficiency). Fuel cell में H₂ fuel है और पानी product।
JEE Q20. The cell constant G* has the dimension of:
(A) Length
(B) Length⁻¹ (1/length)
(C) Length²
(D) Dimensionless
✅ Answer: (B) Length⁻¹ (1/length)
Explanation: G* = l/A = length/length² = length⁻¹ (per metre or per centimetre). Cell constant = l/A।
NEET Q21. In a hydrogen half-cell, the electrode reaction is:
(A) H₂ → 2H⁺ + 2e⁻
(B) 2H⁺ + 2e⁻ → H₂
(C) H⁺ + e⁻ → ½H₂
(D) Both (B) and (C) are correct
✅ Answer: (D) Both (B) and (C) are correct
Explanation: Both representations are equivalent. (B) and (C) both show reduction at SHE. दोनों equivalent हैं — सिर्फ moles का फर्क है।
CBSE Q22. Molar conductivity of a solution INCREASES with dilution because:
(A) The number of ions per mole increases
(B) Ion-ion interactions decrease and ionic mobility increases
(C) The viscosity of the solution increases
(D) The number of moles of solute increases
✅ Answer: (B) Ion-ion interactions decrease and ionic mobility increases
Explanation: At higher dilution, fewer ion-ion interactions → ions move more freely → higher molar conductivity. Dilute solution में ions freely move करते हैं।
JEE Q23. For the reaction 2Ag⁺(aq) + Cu(s) → Cu²⁺(aq) + 2Ag(s), E°cell = 0.46 V. The value of ΔG° is:
(A) −88.79 kJ mol⁻¹
(B) +88.79 kJ mol⁻¹
(C) −44.40 kJ mol⁻¹
(D) +44.40 kJ mol⁻¹
✅ Answer: (A) −88.79 kJ mol⁻¹
Explanation: ΔG° = −nFE°cell = −2 × 96487 × 0.46 = −88,768 J mol⁻¹ ≈ −88.79 kJ mol⁻¹. ΔG° ऋणात्मक = spontaneous।
CBSE Q24. The electrode at which oxidation takes place in an electrolytic cell is called:
(A) Cathode
(B) Anode
(C) Salt bridge
(D) Reference electrode
✅ Answer: (B) Anode
Explanation: In BOTH galvanic and electrolytic cells: Anode = Oxidation always. Galvanic हो या Electrolytic — Anode पर Oxidation।
NEET Q25. Which of the following correctly represents the cell notation for a Daniell cell?
(A) Cu|Cu²⁺||Zn²⁺|Zn
(B) Zn|Zn²⁺||Cu²⁺|Cu
(C) Zn|Cu²⁺||Zn²⁺|Cu
(D) Cu²⁺|Cu||Zn|Zn²⁺
✅ Answer: (B) Zn|Zn²⁺||Cu²⁺|Cu
Explanation: Anode (Zn) on left, cathode (Cu) on right. | = phase boundary, || = salt bridge. Daniell cell notation — Anode बायीं तरफ।
STATE Q26. Specific conductance of a solution DECREASES on dilution because:
(A) The number of ions per unit volume decreases
(B) The ions become less mobile
(C) Temperature decreases with dilution
(D) The solute decomposes
✅ Answer: (A) The number of ions per unit volume decreases
Explanation: On dilution, volume increases but ions are same → fewer ions per unit volume → κ decreases. Dilute करने पर ions per volume कम हो जाते हैं।
CBSE Q27. The process of coating iron with zinc to prevent rusting is called:
(A) Electroplating
(B) Galvanisation
(C) Anodising
(D) Tinning
✅ Answer: (B) Galvanisation
Explanation: Galvanisation = coating iron/steel with zinc. Zinc (E° = −0.76 V) acts as sacrificial anode. लोहे पर जस्ता चढ़ाना = Galvanisation।
STATE Q28. In the electrolysis of dilute sulphuric acid using platinum electrodes, the gas evolved at the cathode is:
(A) Oxygen
(B) Sulphur dioxide
(C) Hydrogen
(D) Sulphur trioxide
✅ Answer: (C) Hydrogen
Explanation: At cathode: 2H⁺ + 2e⁻ → H₂. Hydrogen gas is liberated. At anode: water is oxidised to O₂. Cathode पर Hydrogen gas निकलती है।
NEET Q29. Which of the following has a constant cell potential throughout its life?
(A) Dry cell
(B) Lead storage battery
(C) Nickel-cadmium cell
(D) Mercury cell
✅ Answer: (D) Mercury cell
Explanation: Mercury cell has constant voltage (1.35 V) because no ionic concentrations change during discharge. Mercury cell का voltage जीवन भर constant रहता है।
JEE Q30. The degree of dissociation (α) of a weak electrolyte can be calculated from conductance data using:
(A) α = Λ°m / Λm
(B) α = Λm / Λ°m
(C) α = Λm × Λ°m
(D) α = Λ°m − Λm
✅ Answer: (B) α = Λm / Λ°m
Explanation: Since Λm < Λ°m, α = Λm / Λ°m gives a value between 0 and 1 (degree of dissociation). Degree of dissociation = Λm / Λ°m।
SECTION 2 — Fill in the Blanks
Electrochemistry Fill in the Blanks 25 Questions — CBSE | NEET | State Boards
Q1. In a galvanic cell, oxidation takes place at the _______ and reduction takes place at the _______.
✅ Answer: anode; cathode
Explanation: याद रखो: AN OX = Anode = Oxidation। RED CAT = Reduction = Cathode।
Q2. The standard electrode potential of the Standard Hydrogen Electrode is _______ V.
✅ Answer: 0.00
Explanation: SHE को 0.00 V assign किया गया है — यह universal reference है।
Q3. The relationship between standard Gibbs energy and cell potential is ΔG° = _______.
✅ Answer: −nFE°cell
Explanation: n = electrons, F = Faraday constant (96487 C mol⁻¹), E°cell = standard cell potential।
Q4. E°cell for Daniell cell = E°(Cu²⁺/Cu) − E°(Zn²⁺/Zn) = 0.34 − (−0.76) = _______ V.
✅ Answer: +1.10
Explanation: Positive value confirms the Daniell cell reaction is spontaneous. धनात्मक = spontaneous।
Q5. The Nernst equation at 298 K is: E = E° − (_______ /n) log Q.
✅ Answer: 0.0592 (or 0.059)
Explanation: At 298 K, 2.303RT/F = 0.0592 V. यह constant 298 K के calculations में use होता है।
Q6. A _______ bridge is used to connect the two half-cells in a galvanic cell and maintain electrical neutrality.
✅ Answer: salt
Explanation: Salt bridge में KCl या KNO₃ in agar gel होता है।
Q7. The unit of molar conductivity in SI system is _______.
✅ Answer: S m² mol⁻¹
Explanation: Λm = κ/c → S m⁻¹ / mol m⁻³ = S m² mol⁻¹।
Q8. Kohlrausch’s law: Λ°m = _______ λ°⁺ + _______ λ°⁻
✅ Answer: ν⁺ ; ν⁻
Explanation: ν⁺ and ν⁻ = number of cations and anions per formula unit of electrolyte।
Q9. For strong electrolytes, the variation of molar conductivity with concentration is: Λm = Λ°m − _______.
✅ Answer: A × √c
Explanation: This is the Debye-Hückel-Onsager equation. A depends on electrolyte type and solvent।
Q10. The quantity of electricity (charge) passed is given by Q = _______ × _______.
✅ Answer: I (current in Amperes) × t (time in seconds)
Explanation: Charge (Coulombs) = Current (A) × Time (s) — Faraday’s law में use होता है।
Q11. 1 Faraday = _______ C mol⁻¹.
✅ Answer: 96487
Explanation: Approx 96500 C mol⁻¹ for calculations. यह 1 mole electrons का charge है।
Q12. The electrode at which reduction takes place in an electrolytic cell is called _______.
✅ Answer: cathode
Explanation: Galvanic और Electrolytic दोनों cells में cathode पर reduction होती है।
Q13. The E°cell for a spontaneous electrochemical reaction must be _______.
✅ Answer: positive (greater than zero)
Explanation: E°cell > 0 → ΔG° < 0 → Spontaneous reaction। धनात्मक E° = अपने आप होने वाली reaction।
Q14. The process of depositing a thin layer of a superior metal on a cheaper metal using electrolysis is called _______.
✅ Answer: electroplating
Explanation: Object to be plated = cathode; plating metal = anode। Electroplating में object cathode बनाते हैं।
Q15. The relationship between E°cell and equilibrium constant at 298 K is: E°cell = (0.059 / _______) log Kc.
✅ Answer: n
Explanation: n = number of electrons transferred. Large E° → Large Kc।
Q16. The molar conductivity of a weak electrolyte _______ sharply on dilution.
✅ Answer: increases
Explanation: Dilution → more dissociation → more ions → Λm बढ़ता है तेज़ी से।
Q17. The oxidation half-reaction in the rusting of iron is: Fe(s) → Fe²⁺(aq) + _______ e⁻.
✅ Answer: 2
Explanation: Iron loses 2 electrons to form Fe²⁺. Later Fe²⁺ → Fe³⁺ in rust (Fe₂O₃·xH₂O)।
Q18. In an H₂-O₂ fuel cell, the product formed is _______.
✅ Answer: water (H₂O)
Explanation: 2H₂ + O₂ → 2H₂O. Fuel cell ~70% efficient. केवल पानी बनता है — प्रदूषण नहीं।
Q19. The cell constant G* = _______.
✅ Answer: l/A (length divided by area of cross-section)
Explanation: G* has units of m⁻¹ or cm⁻¹. Determined using KCl solution of known conductivity।
Q20. Conductivity of a solution _______ on dilution, whereas molar conductivity _______.
✅ Answer: decreases ; increases
Explanation: κ घटता है (ions per volume कम), Λm बढ़ता है (ions freely move)।
Q21. The degree of dissociation α of a weak acid = Λm / _______.
✅ Answer: Λ°m
Explanation: Λ°m is calculated using Kohlrausch’s law from strong electrolyte data।
Q22. The Daniell cell can be represented as: Zn | Zn²⁺(1M) _______ Cu²⁺(1M) | Cu.
✅ Answer: || (double vertical line = salt bridge)
Explanation: | = phase boundary, || = salt bridge। यह electrochemical notation है।
Q23. Lead storage battery has a voltage of about _______ V per cell.
✅ Answer: 2
Explanation: Car battery = 6 cells × 2 V = 12 V. Lead battery में 6 cells होते हैं।
Q24. Coating iron with zinc to prevent rusting is called _______, and zinc acts as a _______ anode.
✅ Answer: galvanisation ; sacrificial
Explanation: Zinc is more reactive (E° = −0.76 V), so it corrodes first, protecting iron नीचे।
Q25. At equilibrium in a galvanic cell, the value of E (cell potential) becomes _______.
✅ Answer: zero
Explanation: E = 0 at equilibrium. Then E°cell = (0.059/n) log Kc. No current flows अब।
Electrochemistry Class 12 FAQs
SECTION 3 — Frequently Asked Questions (FAQ)
20 Most Commonly Asked Questions — All Boards
FAQ Q1. What is electrochemistry? (Electrochemistry क्या है?)
Electrochemistry is the branch of chemistry that studies the relationship between chemical reactions and electrical energy. It covers two main things: (1) how spontaneous chemical reactions produce electricity, as in a battery — जैसे हमारे mobile की battery; and (2) how electrical energy is used to drive non-spontaneous chemical reactions, as in electroplating or the production of aluminium.
हमारी daily life में Electrochemistry बहुत important है — phone की battery से लेकर, पानी को purify करने में chlorine बनाना — सब Electrochemistry है।
FAQ Q2. What is the difference between a galvanic cell and an electrolytic cell?
Galvanic Cell: Chemical energy → Electrical energy. Reaction is spontaneous (ΔG is negative). Example: Daniell cell, dry cell (torch की battery).
Electrolytic Cell: Electrical energy → Chemical energy (non-spontaneous reaction). Needs external power supply. Examples: electroplating (चाँदी की परत चढ़ाना), charging a battery, production of aluminium.
सरल भाषा में: Galvanic cell खुद बिजली बनाता है, Electrolytic cell बाहर से बिजली लेता है।
FAQ Q3. What is the role of a salt bridge? (Salt Bridge का क्या काम है?)
A salt bridge serves two very important purposes:
- It completes the electrical circuit by allowing ions to flow between the two half-cells.
- It maintains electrical neutrality in both solutions.
Without a salt bridge, charges would build up in the two beakers and the cell would stop working very quickly. Common salts used: KCl or KNO₃ in agar gel.
उदाहरण: जैसे एक पुल दो शहरों को जोड़ता है — salt bridge दो half-cells को जोड़ता है।
FAQ Q4. What is standard electrode potential? (Standard Electrode Potential क्या है?)
Standard electrode potential (E°) is the potential of a half-cell measured under standard conditions:
— Temperature = 298 K
— Concentration of all ions = 1 mol/L
— Gas pressure = 1 bar
Always measured relative to SHE (assigned 0.00 V). If E° is positive → species is reduced more easily than hydrogen. If E° is negative → hydrogen is reduced more easily.
याद रखो: SHE = 0.00 V reference point।
FAQ Q5. What is the Nernst equation and when do you use it?
Nernst equation is used when concentrations are NOT at standard conditions (not 1 mol/L).
At 298 K: E = E° − (0.0592/n) log Q
where:
E = actual cell potential
E° = standard cell potential
n = number of electrons transferred
Q = reaction quotient
Use it when the question gives ACTUAL concentrations. NEET/JEE में यह equation numerical problems में बहुत use होती है।
Example: अगर [Zn²⁺] = 0.1 M और [Cu²⁺] = 0.01 M है, तो Nernst equation use करो।
FAQ Q6. What is Faraday’s first law of electrolysis?
Faraday’s First Law: The mass of substance deposited at an electrode is directly proportional to the quantity of electricity passed.
Formula: w = (M × I × t) / (n × F)
where:
w = mass deposited (grams)
M = molar mass
I = current (amperes)
t = time (seconds)
n = number of electrons
F = 96487 C/mol
Example: Car battery recharge करते समय, या gold jewellery पर gold plating करते समय — यही law use होता है।
FAQ Q7. Why does molar conductivity increase on dilution? (Molar Conductivity क्यों बढ़ती है?)
On dilution:
— Fewer ions per unit volume → conductivity κ decreases
— But ions are farther apart → fewer ion-ion interactions → ions move more freely → higher ionic mobility
— Since Λm = κ/c (concentration also decreases), the NET result is Λm INCREASES on dilution.
सरल भाषा में: Dilute solution में ions को कम obstacles मिलते हैं, इसलिए वे faster move करते हैं।
Strong electrolyte में: Λm slowly increases (Debye-Hückel-Onsager equation).
Weak electrolyte में: Λm sharply increases (dissociation increases).
FAQ Q8. What is Kohlrausch’s law of independent migration of ions?
Kohlrausch’s law: At infinite dilution, the limiting molar conductivity of an electrolyte equals the sum of individual ion contributions:
Λ°m = ν⁺ × λ°⁺ + ν⁻ × λ°⁻
Example: NaCl के लिए:
Λ°m(NaCl) = λ°(Na⁺) + λ°(Cl⁻) = 50.1 + 76.3 = 126.4 S cm² mol⁻¹
Main use: Weak electrolytes (like CH₃COOH) की Λ°m निकालने के लिए:
Λ°m(CH₃COOH) = Λ°m(HCl) + Λ°m(CH₃COONa) − Λ°m(NaCl)
= 425.9 + 91.0 − 126.4 = 390.5 S cm² mol⁻¹
FAQ Q9. What is a fuel cell? How is it different from an ordinary battery?
Fuel Cell: Reactants (fuel + oxidant) are continuously supplied from outside — it NEVER runs down as long as fuel is provided.
Example: H₂-O₂ fuel cell — H₂ and O₂ react → water + electricity (~70% efficiency).
Ordinary Battery: Fixed amount of reactants sealed inside → runs flat (primary) or needs recharging (secondary).
Fuel cell vs Battery:
— Fuel cell: Continuous supply of fuel → continuous electricity
— Battery: Fixed reactants → limited energy
Future में cars और power plants में fuel cells use होंगी।
FAQ Q10. How does rusting of iron involve electrochemistry? (लोहे पर जंग कैसे लगता है?)
Rusting = electrochemical process. It requires BOTH water and oxygen.
Anodic spots (oxidation): Fe → Fe²⁺ + 2e⁻
Cathodic spots (reduction): O₂ + 4H⁺ + 4e⁻ → 2H₂O
Electrons flow through iron itself. Moisture = electrolyte. Fe²⁺ + O₂ → Fe₂O₃·xH₂O (rust = जंग).
Iron rust is porous — falls off — exposing fresh iron. That is why iron keeps rusting.
Aluminium में Al₂O₃ बनता है जो protective layer है, इसलिए Al नहीं जंग लगता।
FAQ Q11. What are the applications of Kohlrausch’s law?
- Calculating Λ°m of WEAK electrolytes (like CH₃COOH) — cannot be found by direct extrapolation.
- Finding degree of dissociation (α):
α = Λm / Λ°m
- Finding dissociation constant Ka:
Ka = cα² / (1 − α)
- Determining ionic product of water (Kw).
- Solubility of sparingly soluble salts (like AgCl, BaSO₄) — by measuring conductivity of saturated solution.
यह law Electrochemistry का बहुत important tool है।
FAQ Q12. What is the difference between primary and secondary batteries?
Primary Battery:
— Non-rechargeable. Electrode reactions occur only once.
— When reactants are consumed, battery is dead.
— Examples: Dry cell (torch में), Mercury cell (watch में).
Secondary Battery:
— Rechargeable. Reverse current restores original reactants.
— Examples: Lead-acid battery (car में), Li-ion battery (mobile में).
सरल भाषा में:
Primary = Use करो और फेंको।
Secondary = Use करो, recharge करो, फिर use करो।
FAQ Q13. What is cell constant and how is it determined?
Cell constant (G*) = l/A
where l = distance between electrodes, A = area of cross-section.
Unit: m⁻¹ or cm⁻¹.
It is a FIXED property of a particular conductivity cell.
Determination: Fill the cell with KCl solution of KNOWN conductivity (κ_known), measure resistance R.
Then: G* = R × κ_known
Why not measure l and A directly? Because it is impractical for small cells.
Cell constant एक specific cell की property है — solution बदलने से नहीं बदलती।
FAQ Q14. How is the standard electrode potential related to the equilibrium constant?
At equilibrium in a galvanic cell:
— Cell potential E = 0
— Reaction quotient Q = Kc
Substituting into Nernst equation:
0 = E°cell − (0.059/n) log Kc
Therefore: E°cell = (0.059/n) log Kc at 298 K
This is very useful because we can calculate Kc simply from electrode potentials — no experiment needed!
Example: Daniell cell: E°cell = +1.10 V, n = 2
log Kc = (2 × 1.10)/0.059 = 37.16
Kc = 10³⁷·¹⁶ ≈ 2 × 10³⁷ (reaction goes almost completely to products)
FAQ Q15. What is the difference between conductance and conductivity?
Conductance (G):
— Ease of electricity flow through a SPECIFIC conductor
— G = 1/R
— Unit: Siemens (S)
— Depends on size/shape of conductor
Conductivity (κ):
— Conductance of a UNIT CUBE of conductor (1 m × 1 m × 1 m)
— Intrinsic property of the MATERIAL
— Unit: S m⁻¹
— Does NOT depend on size or shape
Relationship: G = κ × A/l
उदाहरण: Different shapes के two copper wires का Conductance अलग होगा, पर Conductivity same।
FAQ Q16. Why is platinum used as an electrode in the Standard Hydrogen Electrode?
Platinum is used in SHE because:
- Chemically inert — does not react with H₂, acid, or products.
- Excellent catalyst for H₂ ⇌ 2H⁺ + 2e⁻ equilibrium.
- High surface area when coated with platinum black.
- Excellent electrical conductor.
सरल भाषा में: Pt reaction में participate नहीं करता, सिर्फ electron transfer का surface देता है — इसलिए यह ideal electrode है।
FAQ Q17. What is meant by limiting molar conductivity (Λ°m)?
Limiting molar conductivity (Λ°m) = molar conductivity at INFINITE DILUTION (concentration → 0).
At this limit:
— All ion-ion and ion-solvent interactions → negligible
— Ions are completely free to move
For strong electrolytes: Plot Λm vs √c → straight line → extrapolate to c = 0 → get Λ°m.
For weak electrolytes: Λ°m CANNOT be measured directly (graph is non-linear). Use Kohlrausch’s law:
Λ°m(CH₃COOH) = Λ°m(HCl) + Λ°m(CH₃COONa) − Λ°m(NaCl)
FAQ Q18. How is galvanisation different from electroplating?
Galvanisation:
— Specifically: coating iron/steel with ZINC to prevent rusting
— Method: hot-dipping in molten zinc OR electrodeposition
— Purpose: corrosion protection
— Zinc = sacrificial anode — even if coating breaks, zinc corrodes FIRST
Electroplating:
— General term: depositing ANY metal on ANY object using electrolysis
— Purpose: decorative AND protective
— Examples: gold plating on jewellery, silver plating on cutlery
सरल भाषा में: Galvanisation = zinc से protection। Electroplating = any metal की coating।
FAQ Q19. What happens to a lead storage battery when it is in use (discharging)?
During DISCHARGE:
Anode (negative, spongy Pb):
Pb + SO₄²⁻ → PbSO₄ + 2e⁻
Cathode (positive, PbO₂):
PbO₂ + SO₄²⁻ + 4H⁺ + 2e⁻ → PbSO₄ + 2H₂O
Both electrodes get coated with PbSO₄. H₂SO₄ is consumed → its DENSITY DECREASES.
State of charge can be checked by measuring H₂SO₄ density with a HYDROMETER.
Recharging: Reverse current → PbSO₄ → Pb + PbO₂ (restored). H₂SO₄ density increases again.
Car की battery की health उसके acid की density से check होती है।
FAQ Q20. What is overpotential, and why is it important in electrolysis?
Overpotential = EXTRA voltage that must be applied beyond the theoretical minimum to make an electrolysis reaction occur at a reasonable rate.
Why does it occur? Some electrode reactions (especially gas evolution) are kinetically SLOW. Even though thermodynamically the reaction should occur at a certain voltage, higher voltage is needed in practice.
Important example: In the electrolysis of a concentrated NaCl solution, water should be oxidised at anode (higher E°). But Cl₂ evolution has a lower overpotential → Cl₂ is produced instead of O₂.
यही reason है कि salt water का electrolysis Cl₂ gas देता है, O₂ नहीं।
Electrochemistry Class 12 Notes + PDF Download + NCERT Solutions Free Download
